Dianisidine
Other names: Dimethyloxybenzidinedianisidine, Dianisidinchlorsulfonat, 3,3'-Dimethoxybenzidine chlorsulfonat, sneezing powder, Ni-geschoss, Niespulver (German: "sneezing powder").
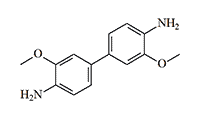
In 1904, a twenty-five-year-old American traveling salesman, Soren Sorensen Adams, who made his living selling dyes and chemical reagents, noticed that a certain substance used in dye manufacturing caused workers to suffer fits of violent, uncontrollable sneezing. The observant young man quickly determined that the culprit was an intermediate compound in the dye synthesis process—o-dianisidine chlorosulfonate. The improvised "sneezing powder" proved an instant sensation among Adams's friends and acquaintances, who would covertly disperse it at parties and revel in the resulting chaos.
 |
 |
| Soren Sorensen Adams (1869–1963)—founder of the S.S. Adams Co. and his "Ca-Choo" sneezing powder. | |
Adams quickly recognized that the novelty had commercial potential. Investing his last available funds, he launched production of the sneezing powder under the trade name "Ca-Choo." The craze swept across America, and the enterprising young entrepreneur went on to found what became the largest manufacturer of gag gifts, novelties, and magic supplies in the country—the S.S. Adams Co. For his outsized contribution to the prank industry, Adams earned the nickname "King of the Gag."
A decade later, the unusual properties of dianisidine came to the attention of a man whose interest in chemistry was anything but playful. Carl Duisberg, director general of Bayer, had been systematically searching for new irritant compounds that might open additional revenue streams for his company. Dianisidine was already in large-scale use at Bayer's dye plants, and Duisberg had observed that workers handling the substance suffered persistent coughing, sneezing, and in some cases respiratory distress. The idea occurred to him: why not deploy this troublesome compound to flush enemy infantry from their trenches and thereby break the deadlock of the attritional trench warfare grinding on at the front? Another German chemist, the future Nobel laureate Walther Nernst, devised a method for loading approximately half a kilogram of dianisidine into a standard 105 mm artillery shrapnel shell. The new munition was designated the Ni-Schrapnell—the prefix taken from the first two letters of Niesen, the German word for sneezing.[1]
 |
 |
| Friedrich Carl Duisberg (1861–1935)—director general of Bayer, and his Ni-Schrapnell. | |
Most senior officers of the German Army regarded chemical warfare with considerable skepticism. One officer—a son of Chief of the General Staff Erich von Falkenhayn—reportedly won a case of champagne by standing for five minutes in the cloud produced by a Ni-Schrapnell detonation without displaying any obvious signs of distress.
Despite these unpromising trials, on October 27, 1914, approximately 3,000 dianisidine-filled shells were fired against British positions—with no discernible effect. The British troops apparently did not even register that they had been subjected to what was, in fact, the first large-scale chemical attack in the history of warfare. The bulk of the irritant was destroyed by the explosive detonation itself, and dianisidine proved far too weak an irritant for effective battlefield use.
Several months later, Duisberg attempted to remedy the situation by submitting an improved version of the Ni-Schrapnell for evaluation—this one filled with a mixture of dianisidine and xylylene dibromide, a lachrymatory agent capable of causing severe eye irritation and tearing. The concept, however, was not pursued further.[3]
Although dianisidine never achieved status as a recognized chemical warfare agent, its irritant properties continued to find application long after the conclusion of World War I. Through the early 1980s, the compound remained an active ingredient in commercially available sneezing powders sold in novelty and joke shops. Its sale was ultimately prohibited after the substance was identified as a probable human carcinogen.[2]