Irritants of Various Groups
Development and Testing of Irritants at Edgewood Arsenal in the 1960s–70s
Most research on irritating chemical agents was conducted in the 1960s. The Vietnam War, where irritants were used on a massive scale, and widespread anti-war protests, which police often dispersed using tear gas, necessitated a new approach to developing irritants for both domestic and foreign use.
Primary research focused on five standard irritating agents (CN, CS, CA, PS, and DM) and three agents that were new at the time — CH, CR, and MPA. The scale of the research is evidenced by the fact that over 1,500 volunteers participated in the trials. The U.S. Chemical Corps searched intensively for new, more effective irritants, but progress was hindered by the lack of a suitable preliminary testing methodology using animals.
In the mid-1960s, Y. Alarie developed a relatively simple method for evaluating the inhalation effects of irritants. By observing changes in the respiratory rate of laboratory mice, it was possible to accurately predict the severity of a substance's irritating effect on humans[2].
Determining effective concentrations of irritants on volunteers at Edgewood Arsenal
Irritants that produce a pronounced irritating effect on humans at concentrations of less than 40 mg•min/m3. The effect was evaluated by the intensity of the pain response in the eyes, nose, throat, and facial skin. The substances are divided into two groups:
- Those not inferior in irritating action to standard chemical agents (marked in blue on the figure).
- Effective at low concentrations but producing a less pronounced effect than CN, CS, and Adamsite (marked in black)[1].
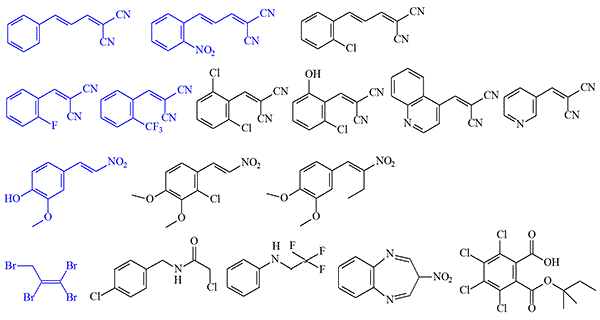
118539 — Lacrimator; causes tearing followed by severe conjunctivitis, salivation, nasal irritation, chest tightness, and coughing. Tested in 1963 (2 people).
123175 — Lacrimator; causes tearing and eye closure for several minutes after exposure ends. Tested in 1965 (8 people).
126312 — Irritant; effect comparable to CS[4].
EA 3365 — Irritant; effect comparable to CS[4].
CS36579 — Causes eye pain, profuse tearing, eye closure and incapacitation lasting several minutes after exposure ends, nasal irritation, and shortness of breath. Tested in 1966 (12 people).
EA 2366 — Causes tearing and conjunctivitis, respiratory tract irritation, shortness of breath, and nausea. Tested in 1966 (2 people).
EA 2097 (Benzylidene malononitrile, CS14632), EA 2214, 118609, 119400, CS36273, CS42985, and CS4332 — Cause feelings of chest tightness and shortness of breath.
EA 2129, EA 3437, CS5635, CS42818, CS24302, CS41458, CS42822, CS42984, CS43010, CS43109 — Upper respiratory tract irritants; cause irritation of the nasal and throat mucous membranes and coughing.Using this methodology, the U.S. Army Chemical Corps screened thousands of chemical compounds annually for irritating properties. The most promising were selected for further human testing at the Edgewood Arsenal research laboratory. Between 1962 and 1972, 123 such substances were selected and tested, but none proved effective enough to replace existing standard irritating agents[3].

Modern plethysmographic setup for measuring respiratory depression (RD50)
In the 1960s-70s, despite extensive research in the USA and UK, no irritant was found that surpassed the physicochemical and toxicological characteristics of the already known and tested CS and CR. Following the end of the U.S. chemical program and stricter requirements for volunteer experiments, the number of trials involving irritants decreased significantly, and the search for new riot control agents virtually ceased. The plethysmographic method for determining respiratory depression (RD50) in rodents remains the primary method for quantifying the effectiveness of sensory irritants today.
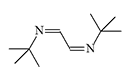
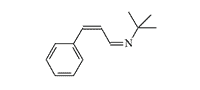
CS 815799 and Azabutadienes
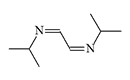
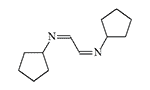
CS 815799. Symmetrical diimines were synthesized in 1970 by J.M. Kliegman and R.K. Barnes during the reaction of glyoxal with the corresponding primary amine. The irritating effect of these substances was discovered by accident when an experimenter suddenly experienced tearing, coughing, and stomach cramps while filtering the tert-butyl derivative in a fume hood. These symptoms disappeared within 5 minutes without visible consequences. The tert-butyl (CS 815799) and isopropyl derivatives exhibit the most pronounced irritating effects[1–3].
 |
 |
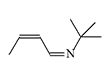 |
| Substance CS 815799 (left) and its isopropyl analogue (right) |
Irritant from the azabutadiene group |
|
Irritating diimines are solid volatile substances with low melting points (40–50ºC).
In 1972, CS 815799 was tested on volunteers at the Edgewood Research Center. The new irritant did not possess outstanding characteristics—a concentration of 50 mg·min/m3 caused moderate eye and respiratory tract irritation. Skin reaction to 1% solutions of CS 815799 was also moderate. Additionally, CS 815799 was noted for a side hepatotoxic effect, though it was completely reversible[6].
Despite the discovered drawbacks, the study of related compounds in the azabutadiene class continued in the USA from 1981–1983 as possible police and/or training chemical agents[4]. Specifically, the Aberdeen Proving Ground (Maryland, USA) investigated the relationship between structure and irritating effect in azabutadienes, finding the highest activity in the isobutyl derivative[5].
RD50 — the concentration of an irritant in mg/m3 that reduces the respiratory rate in mice by 50%. In most cases, such a concentration produces a pronounced or unbearable irritating effect in humans (Y. Alarie, 1973). However, for CS 815799, this pattern does not hold; the substance proved significantly weaker than CS and CR. Inhalation toxicity data for humans regarding the other irritants has not been published.
Diimines
26.00
7.00
2.50
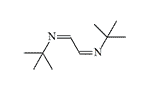
1.20
Azabutadienes
7.5
5.0
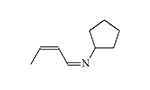
3.56
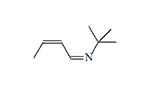
1.8
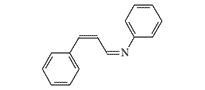
35.0
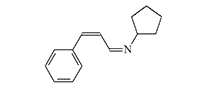
17.0
15.2
For comparison: RD50 for CS is 9.16 mg/m3, and for CR is 2.0 mg/m3.
The Organisation for the Prohibition of Chemical Weapons (OPCW) classifies CS 815799 as a Riot Control Agent (RCA), which is permitted only for law enforcement use. States parties to the Chemical Weapons Convention are required to declare their possession of this substance[7]. Interest in this irritant is likely due to its accessible and inexpensive one-stage synthesis from glyoxal and the corresponding amine.
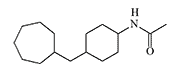
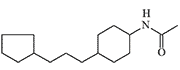
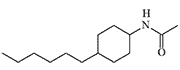
DRC-5593 and Cyclohexylamine Derivatives
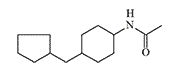
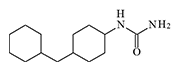
DRC-5593 (N-(4-(cyclohexylmethyl)cyclohexyl)acetamide) was synthesized in the 1960s by Du Pont. The substance possessed an extremely pungent taste and was intended for use as a repellent to deter various animals that cause agricultural damage—from house mice to coyotes. Since Du Pont was a leading contractor for the U.S. Department of Defense in chemical weapons development, the use of this new irritant as a new type of "police gas" was not ruled out.
In 1967–1968, a large number of N-acetylcyclohexylamine derivatives were synthesized, many of which produced pronounced irritating effects in animals. As the new irritants were highly volatile at room temperature, the testing methodology was very simple—animals were simply allowed to sniff the preparation. Holding a piece of filter paper soaked with just 2 mg of the substance to a dog's muzzle was sufficient to cause signs of respiratory irritation, salivation, and extreme agitation. The most active substances tested this way were selected for further inhalation toxicity studies in exposure chambers[1–18].

DRC-5593
The most active irritant obtained, DRC-5593, proved to be an order of magnitude more powerful than CN and CR in animal experiments[25], and its effects lasted longer. Regarding its effect on humans, it is only known that even small concentrations of DRC-5593 vapors cause a severe burning sensation in the nose and chest, coughing, and sneezing[19].
| Substance | RD50 (mg/m3) |
|---|---|
| CR Agent | 2.0 |
 |
2.20 |
 |
4.20 |
 |
7.80 |
| CS Agent | 9.16 |
| Capsaicin | 10.35 |
 |
11.40 |
 |
13.20 |
| DRC-5593 | 44.70 |
 |
58.00 |
Formula |
Minimum |
|---|---|
 |
2 mg |
 |
2 mg |
|
5 mg |
 |
5 mg |
 |
5 mg |
 |
10 mg |
 |
10 mg |
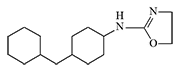
An oxazoline analogue of DRC-5593 was synthesized, likely with the goal of creating a substance with both irritating and "sleep-inducing gas" properties. In addition to irritating the eyes and respiratory system, this compound caused central nervous system depression and muscle weakness in animals[18].
Irritants in this group are considered too toxic for use against humans[1]. Nevertheless, the OPCW included DRC-5593 in a list of 17 potential riot control agents. Besides its use as a "police gas," DRC-5593 may be useful for protecting sheep from coyote attacks or for treating grain[24].
In the early 1970s, Du Pont likely continued developing riot control agents, this time from a group of trifluoromethyl ketone derivatives and diol alcohols.
There is no data on the human toxicity of these irritants; however, it is known that one representative of this group—perfluoropinacol (F3C)2C(OH)—C(OH)(CF3)2—is extremely poisonous; one drop applied to the skin can kill a guinea pig. The Du Pont chemists who synthesized nearly half a kilogram of this substance miraculously escaped harm before it was destroyed[5].
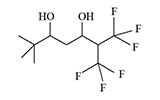
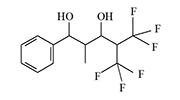
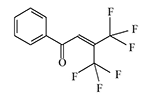
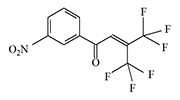
The resulting substances caused eye and upper respiratory tract irritation in animals (mice, rats, monkeys). According to the developers, these irritants "could be used for riot control, rodent control, and in security alarm systems"[1-4].
Substance EA 2542 and Haloacetamides
EA 2542 (2-bromo-N-(2-bromoethyl)acetamide) is another example of an aliphatic compound that, despite its simple structure, possesses a pronounced irritating effect[1]. Volunteer testing of EA 2542 was conducted twice at Edgewood Arsenal—in 1963 and 1969[2].
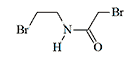
EA 2542
At concentrations of 30–66 mg•min/m3, the substance caused irritation of the eyes, nose, throat, and the area around the eyes. No health consequences were noted in the subjects after exposure to EA 2542[3]. Since EA 2542 is inferior in its irritating effect to modern irritants CS and CR, further study was not pursued.
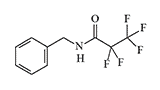
Testing of haloamide derivatives as irritating agents was conducted by the U.S. Chemical Warfare Service shortly after World War I. One of the first irritants in this group, 2'-chloro-2-chloroacetanilide, exerted a pronounced irritating effect on the skin, although it was inferior to known vesicants[3]. In the 1940s, the renowned German military chemist H. Brintzinger worked with substances in this group[4]. In the 1960s–70s, the Edgewood Research, Development and Engineering Center (ERDEC) conducted experiments to evaluate the human irritating effects of at least two substances in this group: 2-chloro-N-4-chlorobenzylacetamide and 2-bromo-N-2-bromoethylacetamide (code EA 2542). Another experimental irritant, N-benzylpentafluoropropionamide, was only half as powerful as CS in animal experiments regarding upper respiratory tract irritation, but its effect on humans is unknown.
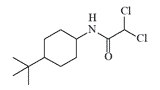
N-(4-tert-butylcyclohexyl)dichloroacetamide proved to be a strong and persistent repellent against dogs, rodents, deer, and other animals[7], but proved too toxic for human use[8].
 |
 |
 |
 |
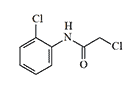
| 2-Chloro-N-(2-chlorophenyl) |
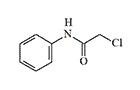
2-Chloro-N-phenyl |
N-Benzylpentafluoro |
N-(4-Tert-butylcyclohexyl) dichloroacetamide |
Chemists working with 2-chloro-N-phenylacetamide encountered its powerful irritating effect. Here is how its effects are described by a person who was unfortunate enough to experience them:
"An incredible irritant. The crystals resemble those of salicylic acid. With it, you first weep, then you'll spend about four hours running (including on walls and ceilings), and as a bonus, you'll itch for about 5 hours. The effect is neutralized with an ammonia solution. It is a serious mistake to try to wash it off with soap and water or alcohol—the effect will only intensify"[5].
Homologues and chlorine derivatives of 2-chloro-N-phenylacetamide, such as 2-chloro-N-phenyl-nonanamide and HPMC, are of much greater interest. In animal experiments, they show activity comparable to or even exceeding that of well-known irritants such as CS and CR[6].
Organic Lead Derivatives

German chemists E. Krause and E. Pohland first encountered the irritating effects of fluorinated trialkyllead derivatives in 1922. Due to the substances' strong irritation of the respiratory organs and eyes, the scientists were forced to wear gas masks while working with them. The slightest inattention resulted in headaches, nausea, and chronic rhinitis. In 1925, two American chemists complained in an article that triethyllead haloacetates caused severe sneezing and were "almost impossible to handle them without suffering considerable inconvenience"[10].
When Great Britain faced a shortage of arsenic at the beginning of World War II—a raw material necessary for the production of agents like Adamsite, diphenylchloroarsine, and Lewisite—British chemists from Cambridge University recalled the suffering of their German colleagues. A group led by B.C. Saunders synthesized and tested a large number of organolead irritants on volunteers, which proved to be just as effective as their organoarsenic competitors. These substances belong to the class of sternutators, or what are now commonly called nasal irritants. They primarily affect the upper respiratory tract, causing excruciating coughing fits, continuous sneezing, severe pain in the throat and gums, and chest tightness. Additionally, some chemists working with these substances complained of burning sensations on the skin of the face and fingers. The lacrimatory (tear-producing) effect of organolead irritants is relatively weak.
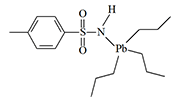 |
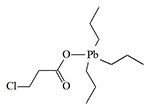 |
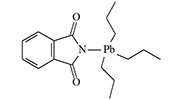 |
Some of the tripropyllead derivatives tested on humans in the 1950s in Great Britain
Meanwhile, across the ocean, American colleagues at the University of Iowa were also working on the synthesis of powerful new irritants based on organic lead compounds[7]. The program was led by Dr. H. Gilman, who had experienced the "charms" of working with organolead in the early 1930s while attempting to create cancer treatments. During the preparation of the drugs, he and his staff coughed, sneezed, wept, and blew their noses continuously. At that time, H. Gilman described the unusual effect of lead compounds on laboratory animals, which, after injection, became "extremely restless, quarrelsome and attack each other at the slightest noise or provocation for over a month"[6]. In total, 84 organic lead compounds were synthesized and tested, but none showed clear advantages over existing irritating agents[7,8].
When the British War Office decided to replace chloroacetophenone with the more effective and safer CS in the late 1950s, it was considered a temporary measure because the effects of CS were not long-lasting enough. Thus, in 1958, lead derivatives were reconsidered. Eight of the most powerful known lead irritants were selected for volunteer testing. These were as effective as the "police gases" of the time—it was practically impossible to endure concentrations of just a few milligrams per cubic meter for more than a minute. Although subsequent tests showed a longer loss of combat capability than after CS exposure, overall, their irritating effect was weaker than that of CS[9].

Another problem with organolead agents was the difficulty of their tactical use—during sublimation, they decomposed into toxic lead or lead-oxide smoke[7].
Tin is in the same chemical subgroup as lead. Between the two World Wars, physiological studies on the systemic toxicity of tributyltin iodide and trimethylpropyltin were conducted in France[12]. During World War II, organohalogen tin derivatives were studied at the University of Chicago’s toxicity laboratory, which worked on chemical weapons development. The compounds studied exerted pronounced irritating and vesicant effects, though concentrations above 1000 mg/m3 were required to achieve toxic effects[13].
Selenium and Tellurium Compounds

Hydrogen selenide (H2Se) is a colorless gas with a foul odor of rotten radishes. The human nose adapts to it quickly and stops sensing it after a while, even though the odor is perceptible at concentrations below analytical detection limits. At concentrations of hydrogen selenide above 5 mg/m3, severe eye and respiratory tract irritation occurs. Without safety measures, further stay in the contaminated atmosphere leads to more serious consequences, such as nausea, vomiting, dizziness, a metallic taste, and an unpleasant garlic breath. Following World War I, hydrogen selenide (G-266), aluminum selenide (G-262), and methyl selenide (G-115) were tested as potential chemical warfare agents at Edgewood Arsenal.
Selenium oxychloride (SeOCl2) possesses even greater toxicity. It penetrates skin well; a few drops of liquid selenium oxychloride on a rabbit’s skin can be fatal. A case is described where 0.005 ml of selenium oxychloride on human skin caused painful swelling and a burn that took a month to heal[1]. Even vapors of this substance can cause redness, swelling, and blistering of the skin[2]. The use of selenium oxychloride as a chemical weapon was not ruled out[3].
During World War II, the U.S. studied the possibility of using selenium oxide (SeO3) as smoke or vapor. It had advantages over another candidate, cadmium oxide, which was as toxic as (or perhaps more toxic than) phosgene. Selenium oxide acted faster and caused respiratory irritation, but was less toxic than cadmium salts. Selenium and cadmium were planned for inclusion in incendiary thermite compositions[7].
In the USSR during the 1930s, work was conducted on selenium mustard gas; however, it was soon discovered that it, like other mustard analogues (fluorine, bromine, selenium), "has no practical value for use in the Red Army"[5].
Many organic selenium compounds have an unpleasant odor perceptible at concentrations as low as 0.00014 mg/m3 (ethyl selenomercaptan).


Diethyl telluride (C2H5)Te. Diethyl telluride was first synthesized by Friedrich Wöhler in 1840. It is a reddish-yellow liquid with a garlic odor, boiling at 138°C, and is insoluble in water. It is flammable, with a risk of spontaneous combustion in air and explosion upon contact with water. In the 1930s–40s, diethyl telluride was studied as a potential chemical agent[3].
In January 1942, the head of the Intelligence Directorate of the Red Army General Staff received a coded message from a residency in Switzerland stating that Germany was accelerating preparations for chemical warfare. According to information obtained from the chief of the anti-chemical protection service at the Swiss War Ministry, "the Germans are manufacturing the following agents in large quantities: mustard gas, phosgene, diphosgene, diphenylarsine cyanide, formyl chloride oxide(?), cacodyl isocyanide, diethyl telluride, nitrosyl fluoride, and ethylarsine chloride"[4].
Organic Antimony and Bismuth Compounds

Antimony and bismuth are in the same chemical subgroup as arsenic and exert similar toxic effects on the human body. In the event of an arsenic shortage, agents based on them could serve as viable substitutes. Thus, in the USSR in 1932, research to find potential chemical agents among organic antimony derivatives began under the leadership of Professor A.N. Nesmeyanov. Since the research was conducted in strict secrecy, little is known about the results. In 1939, the Red Army tested diphenylchlorostibine—a sternutator as irritating as its organoarsenic analogue. Another agent in this group, triphenylantimony, "was considered as part of the chemical weapons system in 1940"[6]. It is only known that this compound causes eye and skin irritation and laryngitis in humans.
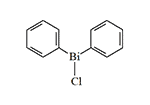
Diphenylchlorobismuth
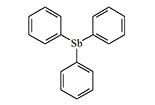
Triphenylantimony
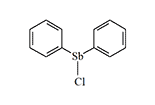
Diphenylchlorostibine
In the UK and USA, the aforementioned Saunders and Gilman synthesized and studied the toxic properties of organobismuth irritants during World War II. Some were even more powerful than World War I sternutators but were ultimately unsuitable for tactical use[11].