Chemical Agent CH (EA 4923)
Military Designations: CH, CHT, 1-MCHT, EA 4923 [US], T4423 [UK], GG [Canada]
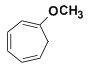
1-Methoxy-1,3,5-cycloheptatriene
Chemical Names: 1-methoxy-1,3,5-cycloheptatriene, methoxycycloheptatriene, 1-methoxytropilidene
Chemical agent CH belongs to a relatively new group of irritants known as algogens—substances that elicit an intense pain response upon contact with skin or mucous membranes, whether as a liquid or vapor. The primary advantage of algogens is their ability to affect targets even when protected by a gas mask. CH vapors affect not only exposed skin but can also penetrate clothing, causing intolerable pain. Notably, unlike other irritants, algogens do not cause chemical skin burns. In addition to CH, substance CR is frequently classified as an algogen, and more rarely, capsaicin and its derivatives.
The precursor to agent CH, 1,3,5-cycloheptatriene, was first synthesized in the 19th century from the alkaloid tropine, from which it derived the name tropilidene. This agent acts as a skin irritant, and its vapors induce lacrimation.
Chemical agent CH (the designation given to 1-methoxy-1,3,5-cycloheptatriene) was first synthesized in the United States in 1962.[5,29] It remains unclear whether its irritant properties were discovered accidentally or through a targeted search for new agents. The synthesis of CH was first described in a 1963 article by Chapman & Borde of Iowa State University,[4] though neither this nor subsequent papers mentioned its physiological effects.[2,3,7] Toxicological studies involving volunteers were eventually conducted at the Edgewood Arsenal Biomedical Laboratory between 1969 and 1971.[10]
Chemical agent CH was evaluated as a promising irritant in the USA (1969–1985),[10,29] the UK (1974–1985),[14] Australia (1971–1972),[29] Canada (1979–1983),[9,17] and Swedish (1982).[30] In Russia, research on CH was conducted at State Scientific Research Institute of Organic Chemistry and Technology (GosNIIOKhT) in 1997[16] and by the "Commercial Security Agency" (2008).[22]
There were also plans to use CH as a training irritant to simulate CBRN (Chemical, Biological, Radiological, and Nuclear) environments. In the USA, agent CS is used to check the fit and integrity of gas masks, while Russia still utilizes the more toxic chloropicrin. However, these irritants are only suitable for testing respirators and cannot evaluate the seal of full-body protective suits. CH is nearly ideal for this purpose; its vapors can penetrate even the smallest gaps, causing burning and pain, which allows for the rapid identification of flaws in protective gear. In the 1990s, the US Marine Corps considered switching to CH for training, but ultimately opted for the more established CS.[23] In the UK, after more than a decade of study, researchers concluded that the advantages of 1-methoxycycloheptatriene were marginal compared to traditional sensory irritants like CS.[14]
In recent years, law enforcement agencies worldwide have increasingly encountered well-prepared groups using professional chemical protection (masks, respirators, gloves), against which standard "police gases" are ineffective. A notable example is the 2004 Beslan school hostage crisis, where the terrorists were equipped with a sufficient number of gas masks.
Mass protests in Turkey (2013), Venezuela (2014), Ukraine (2015), and Hong Kong (2019) also demonstrated high levels of preparation among participants, who used both industrial and improvised respiratory and eye protection to neutralize police special means.
Recently, there has been renewed interest in agent CH from Russian law enforcement. At the 2015 "Non-Lethal Special Equipment" conference, a report noted that "the events of Euromaidan showed that the effectiveness of standard tear gas agents against Instigators of unrest equipped with filtering respiratory protective equipment (PMG-2, PMK, GP-5, GP-7, respirators, etc.) approaches zero." The authors suggested that agent CH could serve as an effective alternative to the current irritants in the Ministry of Internal Affairs arsenal.[6]



During street unrest in Turkey and Venezuela, demonstrators protected themselves from tear gas using simple but effective improvised means. Standard police gases proved useless in these instances.
Physicochemical Properties: A colorless liquid with a boiling point of 117ºC. It degrades when exposed to light and atmospheric oxygen, turning yellowish over time. Antioxidants are required for long-term storage. Its high volatility—8400 mg/m3—allows it to reach intolerable concentrations through natural evaporation in both enclosed spaces and open areas at temperatures above 4ºC. CH is highly soluble in most organic solvents and can penetrate not only skin but also rubber.[10]
| Chemical Agent CH | |
|---|---|
| High concentrations of CS vapor can cause chemical skin burns and blistering. | CH vapors cause only short-term skin redness lasting up to 1 hour. |
| Pyrotechnic mixtures used for dispersal can cause burns, property damage, and fires. | Devices for CH volatilization do not require heating and are simpler and safer to handle. |
| Decontaminating CS from objects and skin requires specialized solutions, consuming time and resources, especially in mass exposure events. | No chemical decontamination is required. Brief ventilation is sufficient to remove residual irritant. |
| CS is easily absorbed by clothing, prolonging its effect on the subject and those around them. This complicates transport in patrol vehicles and can affect others in holding cells. | Due to its high volatility, CH evaporates rapidly from skin and fabric surfaces. |
| Affects only exposed skin. Skin irritation is tolerable. A gas mask provides complete protection. | Penetrates clothing. A gas mask does not protect. |
| Lacrimation stops within minutes; pain subsides within 15 minutes post-exposure. | The effects of CH last for at least 30 minutes. |
Unlike chloroacetophenone (CN) and CS, agent CH does not cause severe chemical burns and does not require thermal dispersal.
The disadvantages of CH include its chemical instability and its complex, expensive synthesis, which makes it difficult to obtain a pure product. Furthermore, the long-term safety of this irritant for humans requires more study.
7-Methoxycyclohepta-1,3,5-triene, an isomer of CH, was first synthesized by Doering et al. in 1954.[2] In 1963, Chapman et al. reported the photoisomerization of the 7-methoxy isomer into the 1-methoxy isomer.[4] In 1974, Japanese chemists discovered that heating the isomer to 450°C results in a quantitative transformation into agent CH.[3]
In 1962, Parham et al. synthesized the ethoxy-homologue of CH starting from cyclohexanone.[1] In 1965, Lindsey & Reese used the same pathway to obtain CH with a 33% yield.[7]
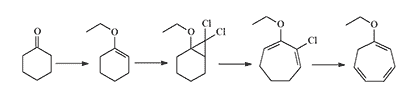
Later, in 1989, this synthesis method was refined in Canada, where adjusting the solvent and catalyst increased the yield of CH to 62%.[9]
the USA, CH was produced via a different method using tropylium tetrafluoroborate as the starting material. This method yielded CH at 92-94% purity with a 70–75% yield.[8]
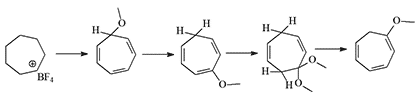
In 1997, the State Scientific Research Institute of Organic Chemistry and Technology (GosNIIOKhT) used this method to synthesize 7-methoxy-1,3,5-cycloheptatriene and several derivatives where the methoxy group was replaced by thio-alcohol, cyano, or malononitrile groups. Instead of tetrafluoroborate, they used the more accessible hexachlorophosphate-chloride, a method developed by Kursanov and Volpin (1957). The resulting substances were liquids with a sharp, irritating odor. Toxicity data were not provided.[16]
Toxicity. According to American researchers, the median incapacitating concentration (ICt50) ranges from 25 to 50 mg·min/m3.[15] In Canadian experiments, the ICt50 was slightly lower, at 11–12 mg·min/m3; notably, no subjects could withstand agent CH vapors at this concentration for more than 3 minutes.[17] If respiratory and eye protection are used, a concentration of 100 mg·min/m3 is required to induce skin pain.
CH is considered safe in that it lacks mutagenic or carcinogenic effects and does not cause long-term damage to the eyes or respiratory tract. Biochemical and clinical analyses taken from volunteers immediately after exposure showed no pathological changes. Compared to pepper sprays, agent CH is ten times less likely to cause allergic reactions.[23] Nevertheless, further research is needed to confirm its safety profile. Animal studies have shown that intravenous doses exceeding 10 mg/kg cause cerebellar cell damage in dogs.[11] Conversely, monkeys tolerate concentrations 2,000 times higher than the human ICt50 without any adverse effects.[10]
Clinical Presentation: Gas masks and standard clothing do not protect against agent CH. Contact with agent CH vapors or solutions results in intense pain and skin redness similar to a first-degree burn, without blistering. The pain response is so severe that individuals are unable to tolerate it for more than a few minutes and are forced to leave the area of contamination immediately. Burning sensations in the eyes and lacrimation do not appear immediately, but rather after about a 40-second delay; however, irritation of the eyes and respiratory tract is generally milder than with other irritants.[15]
If a gas mask is worn, the effects of CH are delayed; after 15 minutes of exposure to vapors, intense burning of the skin occurs under clothing—particularly in the axillae, groin, and sweaty areas. By 20 minutes, the pain becomes entirely unbearable.
Upon moving to fresh air, lacrimation stops within a few minutes, pain subsides within 15–20 minutes, and skin redness disappears after approximately 1 hour.[17]
| Characteristic | Agent CS |
Agent CR |
Agent CH |
|---|---|---|---|
| Volatility at 20ºC (mg/m3) | 0.36 | 0.63 | 8484 |
| ICt50 Median Incapacitating Concentration for humans (mg·min/m3) |
6.9 | 0.15 | approx. 50 |
| LCt50 Median Lethal Concentration (guinea pig, mg·min/m3) |
36,439 | 169,000 | 176,000 |
| LCt50 Median Lethal Concentration (dog, mg·min/m3) |
29,748 | 57,171 | 63,322 |
Data from T. C. Simmons et al. (1976).[10]
Application: Since effective irritating concentrations of agent CH can be achieved through natural evaporation, there is no need for thermal or mechanical aerosol generators. Special formulations containing antioxidants and solvents are preferred. A formulation proposed by Canadian chemists consisted of agent CH, the antioxidant ionol (BHT), and isopentane in a 1:0.5:100 ratio. [17]
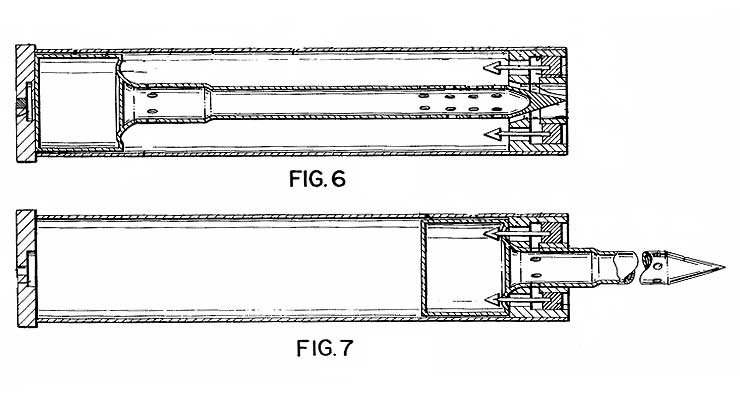
Prototype device for chemical agent CH dispersal in enclosed spaces (Grant, 1986)
Specialized projectiles were developed as prototypes for the rapid pressurized dispersal of CH behind barriers (walls, doors, etc.).[17] Agent CH solutions were also intended for use in "stinging foam" generators to deny access to protected areas by unquthorized personnel.[24] Agent CH can be combined with psychotomimetics of the BZ type to enhance their ability to incapacitate through clothing.[12] Adding a skin-penetration enhancer (DMSO) to CH increases its irritant effect by 1.5 times.[26]
Decontamination is not required, which is a major advantage of CH over other irritants. It does not linger in clothing fabrics, upholstery, curtains, or drapes. Even after indoor application, 30 to 60 minutes of ventilation is sufficient to eliminate its effects. In contrast, removing solid irritants like CN and CS through ventilation alone would take over a year.[25]
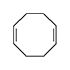
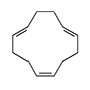
1,5-Cyclooctadiene. A liquid with an unpleasant odor. Vapors cause severe irritation of mucous membranes; skin contact leads to an inflammatory reaction that may result in necrosis. Eye contact causes painful but short-lived conjunctivitis and purulent blepharitis, which may persist for several days.[18] Cyclododeca-1,5,9-triene acts similarly, but the resulting blepharitis is more severe.[19]
 |
 |
 |
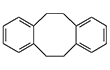 |
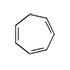
| 1,5-Cyclooctadiene | Cyclododeca-1,5,9-triene | 1,3,5-Cycloheptatriene | 1,2,5,6-Dibenz-1,5-cyclooctadiene |
1,3,5-Cycloheptatriene (Tropilidene; CHT) has a lacrimatory effect but does not cause eye damage. Unlike other cycloolefins, skin irritation occurs without subsequent sensitization.[20]
1,2,5,6-Dibenz-1,5-cyclooctadiene. This substance possesses lacrimatory properties.[21]
EA 4922 — the 3-methoxy isomer of CH—has a similar but weaker effect. The 7-methoxy isomer can cause more serious injuries, such as keratitis or corneal leukoma.[14]