Inhalation Anesthetics
Medieval anesthesiology relied on several competing approaches to pain relief: physical methods (a blow to the head with a heavy object, ice-cold immersion, bloodletting, and carotid artery compression) and pharmacological ones (alcohol, cocaine, mandrake, opium, and hashish). Fortunately for all of us, the advocates of drug-based analgesia ultimately prevailed—largely due to the discovery, in the mid-nineteenth century, of three volatile anesthetics: nitrous oxide (1844), diethyl ether (1846), and chloroform (1847).
Inhalation Anesthetics as Incapacitating Agents
The brilliant minds of the nineteenth century were not content to limit these agents to obstetric analgesia or dental extractions. The first recorded proposal to weaponize anesthetics came from one Joseph Lott, who in 1862, during the American Civil War, suggested mounting pumps on fire engines to spray chloroform over enemy positions. Confederate forces similarly contemplated using chloroform to capture the Monitor, the Union's first ironclad warship.[9]
Both schemes were wildly impractical, requiring enormous quantities of anesthetic to affect open-air targets. More realistic applications emerged for enclosed spaces, where far smaller volumes could achieve effective concentrations.
In 1862, A. Oberndorf proposed incorporating chloroform or ether into bank safe designs as an anti-burglary measure. Any attempt to force or blast the lock would trigger a striker mechanism, rupturing a reservoir of the anesthetizing—or outright toxic—volatile agent.[41]
In 1907, Carl H. Wheaton patented a rather inventive method of neutralizing enemy submarines. His device was designed to pierce a submarine's hull and discharge a sleep-inducing gas into the engine room; once the crew was incapacitated, the vessel could be boarded and taken without a fight.[40]
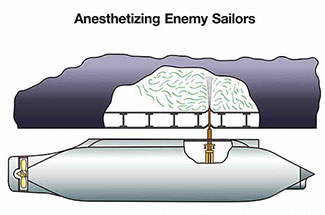
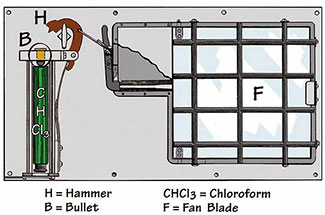
Chloroform and ether were quickly adopted by intelligence services and criminal actors alike. However, both agents have a sharp, distinctive odor that makes covert administration nearly impossible. In practice, clandestine use has relied on what medicine calls Rausch narcosis—a saturated cloth pressed firmly over the victim's face until unconsciousness sets in.
Nitrous oxide (N2O) (commonly known as laughing gas) is a colorless, non-flammable gas with a faint, mildly sweet odor. It is considered one of the safest anesthetic agents in clinical use. While nitrous oxide is nearly ideal for medical anesthesia, it is wholly impractical for weaponization: its physical and chemical properties require extremely high atmospheric concentrations to produce narcosis, making battlefield or covert deployment essentially unfeasible.
Another candidate as a "knockout gas" of that era was carbonyl sulfide (S=C=O)—a colorless, odorless gas with a boiling point of −50°C. The noted military toxicologist V. Lindemann cited it as a potential narcotic agent in a monograph published in the 1930s.[37] However, its anesthetic potency proved too low while its toxicity was far too high for practical use.[38]

In the French film The Hunter Will Get You, the protagonist uses dental nitrous oxide canisters to render criminals unconscious inside a van.
In 1932, a small-scale study was conducted at Edgewood Arsenal evaluating both the lethal and narcotic concentrations of seven anesthetics known at the time.[63] All of them proved wholly unsuitable for combat application.
The first major advance in inhalation anesthetics came with the introduction of Halothane into clinical practice in 1956—marketed in the Soviet Union as Ftorotan. Halothane is a volatile, colorless liquid with a characteristic, not unpleasant odor; its vapors are neither flammable nor explosive. The threshold concentration reliably producing loss of consciousness is 1% by volume, while half that dose—0.5%—is ineffective even after five minutes of inhalation.[48] Offering a number of advantages, including precise depth control and smooth induction and emergence, halothane gradually displaced the older anesthetics from operating rooms.
Halothane never quite achieved the status of "anesthetic of choice," however, as it occasionally caused severe hepatotoxicity. Nevertheless, owing to its low cost, it has remained one of the most widely used general anesthetic agents worldwide. Halothane also finds limited industrial application as a propellant, marketed under the designation Freon 123B1.
More promising proved to be enflurane (1967), and especially its isomer isoflurane (1981), which is still regarded as one of the premier inhalation anesthetics in use today. Isoflurane's principal advantage is a near-complete absence of hepatotoxicity. The more modern sevoflurane (1990) has a pleasant odor, which makes mask induction straightforward even in pediatric patients. Unfortunately, the newest-generation agents—desflurane (1992) and sevoflurane—remain relatively limited in availability.
| Name and synonyms |
Chemical formula | MAC (%) | Anesthetic concentrations (vol.%) | Anesthetic potency (N2O = 1) |
|
|---|---|---|---|---|---|
| Induction | Maintenance | ||||
Halothane |
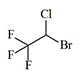 |
0.74 | 2–3.5 | 0.5–1.5 | 142 |
Methoxyflurane
Pentran, |
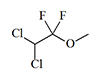 |
0.26 | 2 | 0.8–1 | — |
Enflurane
Efrane, |
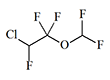 |
1.68 | 2–8 | 1–3 | 62.5 |
Isoflurane
Forane, |
 |
1.15 | 2.5–4 | 1–2.5 | 91 |
Sevoflurane
Petrem, |
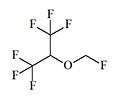 |
2.05 | 5–8 | 0.5–3 | 51 |
Desflurane Suprane |
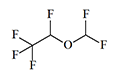 |
6.0 | 4–11 | 2–6 | 17.5 |
MAC (minimum alveolar concentration) is the alveolar concentration of an inhalation anesthetic at which 50% of patients show no motor response to a standard stimulus (skin incision).
In the mid-1950s, American intelligence took an interest in halothane. The CIA, acting through one of its numerous contractors, procured approximately 250 kg of halothane for internal use at a cost of $12,000.[11]
Prior to the 1980s, open-source Western military toxicology literature contained virtually no references to potential applications of inhalation anesthetics. In 1984 and again in 1989, however, the U.S. Department of Defense funded programs aimed at discovering "new quick-acting physically immobilizing compounds which are effective by inhalation; and to synthesize and evaluate potent analgesics and volatile anesthetics to optimize dissemination techniques/hardware to satisfy program requirements."[14,64] In 1989, at the Conference on Chemical Defense Research, the U.S. Army Chemical Research, Development & Engineering Center (CRDEC) presented a report on the pharmacokinetics of inhalation anesthetics as potential incapacitating agents.[43]
Between 1988 and 1990, CRDEC, in collaboration with the Medical College of Virginia, conducted research into the structure–activity relationships of anesthetics in the fluorinated cycloalkane series.[66]
Some promise for improved safety margins was seen in cycloalkanes (cyclobutane, cyclopentane, and cyclohexane) bearing fluoromethyl substituents—CF3, CF2H, and CH2F. Between 1990 and 1991, CRDEC contracted with Iowa State University for the synthesis of trifluoromethylcyclohexanes.[67] In 1993, chemists at Edgewood Arsenal developed their own synthetic methodology for fluoro- and trifluoromethyl-substituted cyclohexane derivatives.[68]
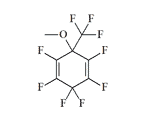
Four of the compounds prepared at ERDEC were forwarded for further pharmacological evaluation; the results were never published.
In 2001, the Applied Research Laboratory at Pennsylvania State University assessed the potential military utility of isoflurane and sevoflurane.[1,2] In CRDEC documents, methoxyflurane, alongside other agents, is listed as a probable chemical incapacitant.[12] Information on inhalation anesthetics was also featured in one of the authoritative databases on chemical and biological weapons—Chemicals and Agents of Biological Origin (CABO)—used by the U.S. Department of Defense and the intelligence community.
Limitations of Inhalation Anesthetics as Non-Lethal Chemical Agents
The primary criterion for any incapacitating agent is a high margin of safety, which for inhalation anesthetics is expressed as the anesthetic index—the ratio of the median lethal concentration (LC50) to the median effective concentration (AC50). The higher this LC50/AC50 ratio, the lower the risk of fatal overdose during deployment. Unfortunately, inhalation anesthetics perform poorly by this measure: for all commercially available agents, the LC50/AC50 ratio does not exceed 5. By comparison, the incapacitating dose of the psychochemical agent BZ is 40-fold lower than its lethal dose, while for LSD the margin is at least 250-fold.
More than a century and a half has passed since the discovery of ether and chloroform. In that time, hundreds of compounds capable of producing narcosis in humans and animals have been identified and characterized. Many of the shortcomings of the early anesthetics have been overcome: general anesthesia has become considerably safer and more controllable, and adverse effects have grown less frequent. Yet one property of volatile anesthetics has remained essentially unchanged—their potency. Most modern inhalation anesthetics are less potent than the chloroform discovered in the nineteenth century. Contemporary incapacitating and calmative agents, by contrast, produce incapacitation at concentrations thousands of times lower than those required for inhalation anesthesia. Accordingly, volatile and gaseous anesthetics have only three plausible applications in this context:
- In combination with CNS depressants with synergistic activity: benzodiazepines, ketamine, α2-adrenomimetics (but not opioids);
- In confined spaces with minimal air recirculation (enclosed rooms, aircraft or vehicle cabins, and similar environments);
- As a solvent or carrier vehicle for other temporarily incapacitating agents (irritants, calmatives, psychochemical agents).
The most favorable combination is generally considered to be an anesthetic paired with an α2-adrenomimetic such as medetomidine. Animal experiments have demonstrated that medetomidine reduces the MAC of halothane by as much as 90%.[50] Between 2010 and 2013, researchers at Imam Hussein University in Iran developed an aerosol incapacitant formulation based on sevoflurane, medetomidine, and ketamine.[13] This same combination is routinely used in veterinary medicine for anesthesia across a wide range of species—from domestic poultry to tortoises and crocodilians.[51]
A significant drawback of most inhalation anesthetics is their sharp, pungent odor, which severely compromises covert application. Notable exceptions include xenon, nitrous oxide, and the poorly characterized Heptafluorocyclopentane 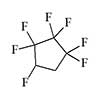 (HFCP).[18] Anesthetics that are odorless or have a mild, pleasant scent—such as cyclopropane and sevoflurane—also exhibit the fastest onset of action: loss of consciousness can occur within one or two breaths. Attempts to render a person unconscious by pressing an ether- or chloroform-soaked cloth to the face, by contrast, require far longer than typically depicted in film.
(HFCP).[18] Anesthetics that are odorless or have a mild, pleasant scent—such as cyclopropane and sevoflurane—also exhibit the fastest onset of action: loss of consciousness can occur within one or two breaths. Attempts to render a person unconscious by pressing an ether- or chloroform-soaked cloth to the face, by contrast, require far longer than typically depicted in film.
Use of "Knockout Gases" in the Soviet Union
The prospect of capturing cities without firing a shot and laying entire armies to sleep was sufficiently alluring that as early as 1924, the government of the young Soviet state approached Professor A. E. Chichibabin and invited him to participate in research on "narcotic agents" for military purposes. Negotiations over terms and compensation broke down, and the eminent Russian chemist declined to take part.[36] Research in this direction nonetheless continued actively within the USSR. In 1934, plans were drawn up at Chemical Plant No. 1 in Moscow for the "synthesis of substances potentially possessing narcotic properties."[35]
In the second half of the 1940s, a genuine underground war was under way in western Ukraine between Ukrainian nationalist insurgents—the Ukrainian Insurgent Army (UPA) and the Organization of Ukrainian Nationalists (OUN)—and Soviet military and state security forces. To sustain their clandestine operations, the insurgents constructed and concealed an extensive network of underground hideouts and bunkers. These dugouts were meticulously camouflaged and typically situated in unusual or hard-to-reach locations: attics, basements, wells, cemeteries, and abandoned buildings. Most commonly, however, they were dug in dense forest, with entrances carefully hidden from view. In the Lviv region alone, more than 2,500 such shelters were discovered in the space of just two months.[44]

Soviet security personnel negotiating with Ukrainian insurgents through a ventilation shaft of an underground bunker (western Ukraine, 1945).
A critical feature of these shelters was a ventilation shaft, which also served as a chimney flue—and which most often betrayed a hideout's location. Once a discovered bunker had been surrounded by troops, narcotic gas was pumped in through the ventilation opening from small portable cylinders. Initially the agent used was designated "Neptune-7/93"; in the early 1950s it was superseded by a more effective agent called "Typhoon." Nothing is known about the effects of these gases, let alone their composition. "Typhoon" was almost certainly odorless and acted quietly and rapidly, leaving occupants no opportunity to resist or detonate explosives. In at least one documented incident, insurgents who heard the hiss of a gas cylinder being opened understood what was coming and took their own lives.[44]
Inhalation narcotics were brought back into public discussion when press reports emerged suggesting that the "narcotic gas" used during the 2002 Moscow theater hostage crisis at the Dubrovka Theater Center may have included Halothane. This conclusion was reached by Professor T. Zilker, a Munich physician, after reviewing blood and urine analyses from two of his German compatriots who had been affected by the gas. Whereas halothane's presence in one case could conceivably be attributed to accidental exposure during mechanical ventilation at the hospital, in the second case the only plausible source was the gas deployed during the assault.[21]
J. Miller, a pharmacologist at the University of South Carolina, drew attention to two additional indirect indicators pointing to halothane. Halothane is a volatile liquid with a sweetish odor whose vapors have a bluish-gray tint—both features that recur in the accounts of survivors. The distinctive smell of halothane was also identified by one of the hostages who was himself an anesthesiologist at a Moscow hospital.[30]
Halothane was manufactured in the USSR at the Altaikhimprom plant in Slavgorod. In 1977, plant management received a directive to increase annual Halothane (Ftorotan) production from 40 to 200 metric tons. The prominent Russian environmentalist Lev Fedorov argued that such a five-fold increase could not be explained by any projected growth in surgical volumes and must therefore have been connected to the compound's use in an incapacitant formulation.[47]
Halothane could not, however, have been effective as a standalone agent. According to calculations by Professor Van Aken of the University of Münster, rapidly rendering the number of people in a space the size of the Dubrovka concert hall unconscious would have required the rapid vaporization of 3,000 to 5,000 liters of halothane—a wholly impractical quantity.[3,4]
A more plausible interpretation holds that halothane served as a solvent for carfentanil and remifentanil, both of which were detected in clothing samples and biological specimens from affected individuals. Halothane had previously been explored as a carrier solvent for another incapacitant: in the late 1960s in the United Kingdom, it was investigated as a propellant for the irritant agent CR in personal protection devices.[15] Moreover, inhalation anesthetics and fentanyl-class opioids are known to potentiate one another: in combined use, remifentanil has been shown to reduce sevoflurane requirements during surgery by as much as 95%.[31]
On the other hand, halothane at sub-anesthetic concentrations depresses both respiratory rate and tidal volume,[48] while remifentanil causes acute respiratory depression in 1–10% of cases.[49] Both agents also induce bradycardia and hypotension. There is therefore no reason to assume that their combined use would produce synergism exclusively in anesthetic potency while sparing the potentially lethal effects on the cardiovascular and respiratory systems. This view is shared by T. Stanley, who collaborated with CRDEC on research into fentanyl-class incapacitants. He has argued that the co-administration of opioids with agents that enhance their action—including inhalation anesthetics—substantially narrows the therapeutic index of the opioids.[32]
The Most Potent Inhalation Anesthetic Agents
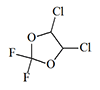
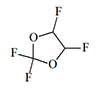
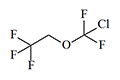
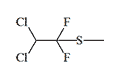
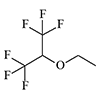
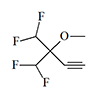
Thiomethoxyflurane—which surpasses methoxyflurane in potency by a factor of 7.5—currently holds the distinction of being the most potent inhalation anesthetic known, with a MAC of just 0.035%.[5] Despite this remarkable potency, thiomethoxyflurane has found no clinical application, for three reasons: an unpleasant odor, low volatility, and considerable toxicity. Somewhat less potent is the cyclic compound dioxychlorane, with a MAC of 0.11%.[6] Dioxyflurane and Compound 485 are also regarded as highly potent anesthetics.
 |
 |
 |
 |
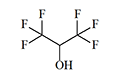 |
| Dioxychlorane | Dioxyflurane | Compound 485 | Thiomethoxyflurane | Hexafluoroisopropanol |
Hexafluoroisopropanol, a metabolite of sevoflurane, produces anesthesia in mice at a concentration of 44 parts per million (MAC = 0.0044%). The corresponding MAC in humans is likely even lower—approximately 25 ppm, or roughly 300 mg/m3—a toxicity level comparable to that of chemical warfare agents from the Second World War era.[10] Any clinical application of hexafluoroisopropanol is, however, precluded by the fact that it is a potent irritant and can cause irreversible ocular damage.[39]
Compound |
AC50 |
LC50 |
AI |
Ref. |
|---|---|---|---|---|
| Isoflurane | 1.28 | 3.2 | 2.5 | [22] |
| Chloroform | 0.78 | 2.6 | 3.3 | [22] |
| Sevoflurane | 2.13 | 7.34 | 3.46 | [23] |
| Diethyl ether | 3.2 | 11.2 | 3.5 | [65] |
| Halothane | 0.78 | 2.74 | 3.5 | [24] |
 |
1.80 | 7.87 | 4.4 | [25] |
| Methoxyflurane | 0.51 | 2.28 | 4.5 | [24] |
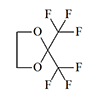 |
0.50 | 2.38 | 4.7 | [26] |
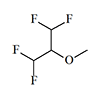 |
1.40 | 10.3 | 7.3 | [27] |
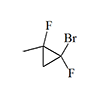 |
0.5 | 5 | 10 | [28] |
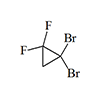 |
0.25 | 3 | 12 | [29] |
 |
0.35 | 5.55 | 16.1 | [24] |
LC50—median lethal concentration causing death in 50% of animals (vol.%);
AI—anesthetic index (LC50/AC50); the higher the value, the greater the margin of safety.
Hundreds of narcotic compounds have now been studied and characterized. They span a wide range of physical forms—gases and volatile liquids alike—and some exhibit considerable potency and rapid onset, are colorless, and have no detectable odor. Narcotic gases are plentiful. Safe ones do not exist.