Convulsants II. Silatranes

Academician Mikhail Voronkov (1921–2014), who first described the potent physiological effects and toxicity of silatranes
Organosilicon Compounds as Potential Chemical Warfare Agents
The staggering success of German chemical warfare during World War I compelled the Allied powers to launch an extensive search for new, highly toxic agents. In post-war France, this momentum led researchers to focus on organoelement compounds, investigating derivatives of lead, tin, selenium, antimony, mercury, magnesium, and silicon. Among the organosilicon compounds studied during this period, some exhibited distinct irritant and vesicant properties. Ultimately, however, their incapacitating effects were deemed insufficient for military adoption.
The investigations also uncovered a more insidious side to certain organosilicons: their ability to strike the central nervous system. Beyond merely triggering eye irritation, trialkylmercaptosilanes — with the general formula HSi(S–Alk)3 — left researchers struggling with bouts of drowsiness and debilitating headaches.[56]
Following World War II, volatile silicon compounds were scrutinized in the Soviet Union as potential chemical agents capable of igniting gas mask filters (charcoal beds). Ultimately, however, preference was given to the more toxic phosphine (PH3).[48]
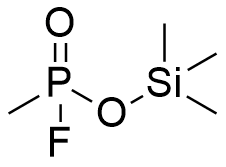
In 1953, a joint British-Australian team of chemists at the Chemical Defence Experimental Establishment (CDEE) investigated the toxicity of a series of organosilicon compounds designed to mimic the structure of "nerve gases." Their findings were discouraging: they concluded that "the low toxicity of the few silicon analogues of the organophosphorus compounds prepared and tested, does not give great hope for finding highly active compounds in this field."[46] Earlier promising toxicological assessments of silicon-based nerve gas analogues — such as Et2SiF(i-PrO), synthesized by their colleagues in the U.S. Army Chemical Corps — also failed to hold up under rigorous testing.[57]
In the USSR, however, the outlook remained far more optimistic. In 1958, the prominent Soviet military chemist L. Soborovsky synthesized trialkylsilanol analogues of Sarin.[47] While no data regarding their toxicity was released to the public, the Soviet leadership was clearly intrigued. This interest culminated in the USSR’s Seven-Year Plan for 1959–1965: the section titled "Prospecting for New, Highly Toxic Agents" included a strategic mandate for the "synthesis of new highly toxic substances—acting both via inhalation and through the skin—within the classes of organophosphorus compounds containing sulfur, nitrogen, silicon, boron, and other elements."[49]
 |
 |
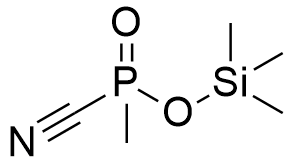 |
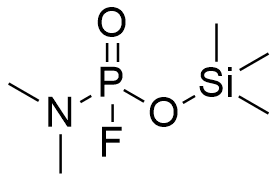
| Si-Sarin | Si-FluoroTabun | Trimethylsilyl Methylphosphonocyanidate |
In his 1994 monograph, Major General N. Antonov—the former commander of the Shikhany military chemical facility—identified derivatives containing silicon atoms in their radicals as being among the six most toxic groups of fluorophosphonates.[64] While the specific identities of these substances were not disclosed, specialized literature contains references to the trimethylsilyl analogues of Fluorotabun and Methylphosphonocyanidate.[65]
Silatranes
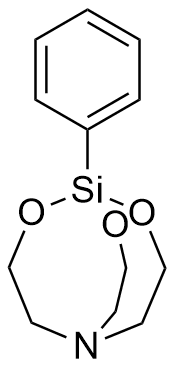
The bicyclic organosilicon compounds that would later be known as silatranes were first synthesized around 1960 by two independent groups of American chemists.[74,75] However, it was in 1962 that their physiological potential caught the attention of the laboratory of the renowned Soviet chemist Mikhail Voronkov (1921–2014). The new substances proved to be extremely toxic to mammals—roughly twice as toxic as strychnine—while remaining puzzlingly harmless to frogs, microbes, and fungi.[8]
Silatranes function as non-competitive blockers of the chloride channel within the GABAA receptor complex. By binding to the picrotoxinin site of the receptor, silatranes effectively "lock" the ion channel, preventing the flow of chloride ions into the neuron. This results in a sharp suppression of inhibitory processes in the central nervous system, leading to uncontrolled neuronal excitation. Externally, this manifests as severe convulsions.
As the West German chemist U. Wannagat later described it: "The discovery made by the Russian scientist M. Voronkov struck like a bolt of lightning: silatranes, emerging from a group of seemingly 'harmless' organosilicon compounds, proved to be highly toxic. A single crystal the size of a matchhead is enough to kill a human being."[8]
Mikhail Voronkov maintained that the toxic properties of silatranes came as a complete surprise to him. However, historical scrutiny suggests a more nuanced reality; Voronkov’s biography reveals that much of his career was, directly or indirectly, intertwined with military chemistry. At the beginning of World War II, Voronkov served as the head of a battalion's chemical service. Late in the war, he joined the Institute of Organic Chemistry of the USSR Academy of Sciences in Kazan—the very site where Soviet Sarin had been synthesized in 1943, and where research into other organophosphorus chemical agents only accelerated in the post-war years.
 |
 |
 |
 |
| Phenylsilatrane LD50 – 0.33 mg/kg (mice, ip)[31] |
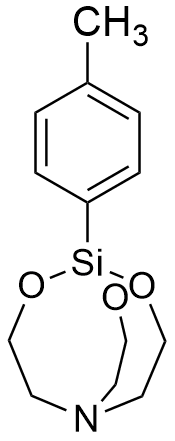
p-Tolylsilatrane LD50 – 0.2 mg/kg (mice, ip)[31] |
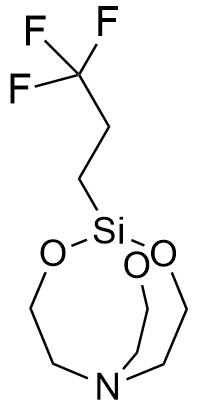
1-(3,3,3-Trifluoropropyl)silatrane LD50 – 0.2 mg/kg (mice, ip)[77] |
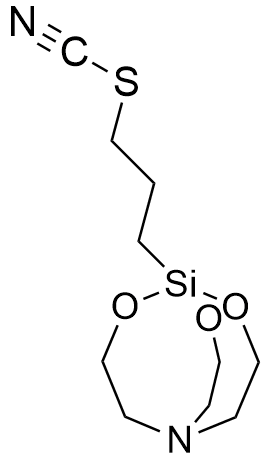
1-(3-Propylthiocyanate)silatrane LD50 – 0.2 mg/kg (mice, ip)[72] |
Since 1961, Voronkov has headed a laboratory at the Institute of Organic Synthesis of the Latvian SSR Academy of Sciences. This institution was a known participant in the development programs for new non-lethal chemical agents[50] and was long suspected of fulfilling clandestine orders for the KGB and the GRU (Soviet Military Intelligence).[51]
In 1970, in recognition of his outstanding achievements in organosilicon chemistry, Voronkov was appointed head of the Irkutsk Institute of Organic Chemistry. Under his leadership, the laboratory synthesized "many other highly toxic silatranes containing hydrocarbon substituents with nitrogen, phosphorus, sulfur, or fluorine atoms attached to the silicon atom."[8] Notable examples include 1-(3,3,3-Trifluoropropyl)silatrane[77] and 1-(3-Propylthiocyanate)silatrane; both compounds exhibit an LD50 of 0.2 mg/kg (mice, ip).[72] While most silatranes cause death within a window of several to tens of minutes, high doses of the 3,3,3-trifluoropropyl derivative induce near-instantaneous fatality.[96]
In light of this, it is telling that when Mikhail Voronkov was asked whom he considered his mentors, he cited Academicians Alexey Favorsky, Nikolay Zelinsky, and Vladimir Ipatieff. All three were prominent members of the Interdepartmental Committee on Chemical Warfare (Mezhsovkhim)—the very organization responsible for preparing the Soviet Union for chemical conflict with the capitalist West.[52]
The United States watched Voronkov’s toxicological work on silatranes with growing concern. A classified 1984 CIA report stated that the Soviets were investigating a number of specific compounds “which appear to have considerable potential as BW [biological warfare] agents." According to the document, these included “silicon-containing and organofluorine compounds... marine neurotoxins and blue-green algal neurotoxins."[69]
1-Phenylsilatrane was a component of South Africa’s chemical weapons program and was intended for the targeted assassination of political dissidents. Experiments with this compound commenced in 1987, beginning with toxicological trials on rats and baboons. In one such experiment, three baboons were administered a 1 mg/kg intramuscular dose of phenylsilatrane; all three animals suffered muscle spasms and disorientation within five minutes. After twelve minutes, they continued to exhibit spasms alongside acute respiratory distress. All subjects died from asphyxiation within fifteen minutes.[65]
Between 1987 and 1988, a London-based agent of the South African Civil Cooperation Bureau (CCB)—a government-sponsored "death squad" during the apartheid era—was provided with a toxin intended for foreign volunteers who worked covertly to assist the African National Congress (ANC). To administer the poison, the agent was issued specialized screwdriver equipped with a syringe-like mechanism hidden within the handle. A single stab with the device would eject a lethal dose of liquid poison into the victim. Ultimately, however, no opportunity arose to carry out the assassination. It is suspected that the substance intended for these operations was 1-phenylsilatrane.[65]
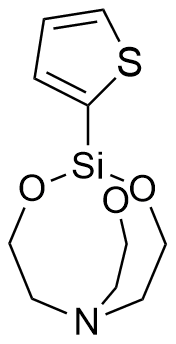
Thienylsilatranes are among the most intriguing members of this organosilicon family, as they exhibit psychotropic activity at doses up to 1,000 times lower than their lethal threshold. Interestingly, the two isomers produce opposing effects: 1-(2-thienyl)silatrane acts as a stimulant, while 1-(3-thienyl)silatrane induces sedation, suppressed orientation reflexes, reduced locomotor activity, and skeletal muscle relaxation. Both isomers impair coordination in mice at a remarkably low dose of 0.0016 mg/kg (i.p.); notably, the LD50 for 1-(3-thienyl)silatrane is 1,125 times higher than its effective dose.[72] Compounds with such potent biological activity were almost certainly evaluated as potential incapacitating agents. It is no coincidence that in 1978, the Swedish National Defence Research Institute (FOA) conducted specialized studies into the toxicity mechanisms and potential therapies for 1-(2-thienyl)silatrane poisoning.[71]
 |
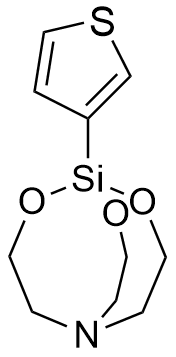 |
| 1-(2-Thienyl)-silatrane LD50 – 0.3 mg/kg (mice, ip)[72] |
1-(3-Thienyl)-silatrane LD50 – 1.8 mg/kg (mice, ip)[72] |
Treatment of Poisoning The primary antidotes for the convulsive effects of silatranes are GABAA receptor agonists. Specifically, phenobarbital has been shown to increase the LD50 of 1-(2-thienyl)silatrane by a factor of 13, while diazepam provides a more modest twofold increase.[71]
Other Toxic Organosilicon Compounds
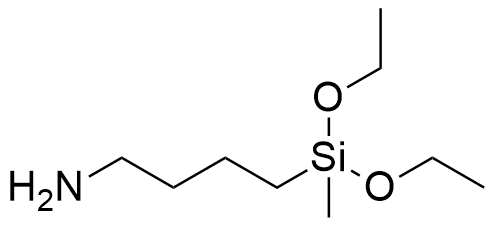
Mikhail Voronkov recalled that his impetus for studying biologically active silatranes was an early report regarding the unusually high dermal toxicity of (4-aminobutyl)diethoxymethylsilane.[31,60] Some sources claimed—likely erroneously—that this substance possessed extraordinary percutaneous toxicity, with a median lethal dose (LD50) of just 0.045 mL/kg when applied to the skin of rabbits.[60]
 |
 |
| Diethoxymethyl(4-aminobutyl)silane LD50 – 0.045 mL/kg (rabbit, skin)[60] |
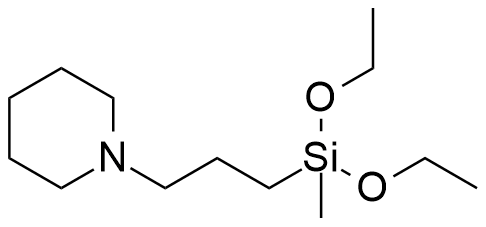
Diethoxymethyl(3-piperidinopropyl)silane LD50 – 6.6 mg/kg (mice, ip)[31] |
Voronkov’s laboratory subsequently synthesized and evaluated twenty structural analogues; however, even the most potent among them failed to match the toxicity levels reported for the original compound.[31]
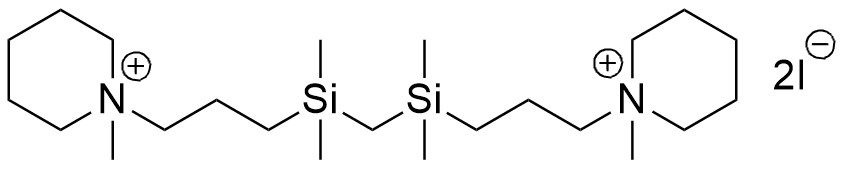
In the mid-1970s, West German chemists Reinhold Tacke and Ulrich Wannagat synthesized silicon analogues of the muscle relaxants suxamethonium and Decamethonium. The latter had previously been evaluated in the United States as a potential non-lethal chemical agent; however, it failed to find either military or clinical use due to its narrow therapeutic index—even a slight overdose could induce fatal respiratory arrest.[40,84] Their organosilicon counterparts proved to be significantly more potent, exhibiting up to 30 times the toxicity of Decamethonium and inducing paralysis in mice at doses as low as 0.07 mg/kg. The authors noted that these compounds appeared to be "the most toxic organosilicon compounds described in the literature up to now."[73,76]
 |
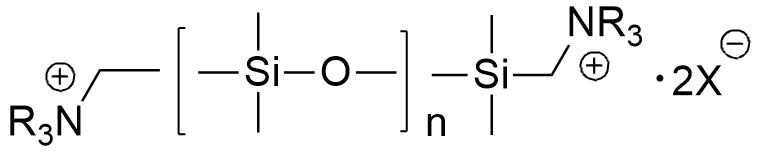 |
| Compound Tacke & Wannagat LD50 – 0.32 mg/kg (mice, ip)[73] |
Compound Voronkov |
Naturally, Mikhail Voronkov could not overlook substances of such extreme biological activity. His laboratory subsequently synthesized several highly toxic structural analogues of Decamethonium, further cementing the status of organosilicon derivatives as a formidable class of synthetic poisons.[85] The era of regarding silicon as a physiologically "inert" element had come to a decisive end.
Despite their high biological activity, silatranes were never seriously considered for development as standard chemical warfare agents (CWA). Compared to V-series or G-series nerve agents, the toxicity of silatranes is relatively moderate, making them inefficient for large-scale military use. Furthermore, their physical form as crystalline solids complicates their "weaponizability"; while some derivatives, such as alkylsilatranes, exhibit significant stability against hydrolysis, the logistical challenges of effective aerosol dispersal and the lack of tactical advantages over established agents relegated them to the periphery of chemical weapons research. Their utility remained confined to specialized clandestine scenarios where their unique forensic signature and specific mechanism of action were of more value than raw lethality.